MacKenzie, and Emilio Segrè, who bombarded. The latest market analysis by Technavio predicts the cesium market will grow by 1.66 thousand MT between 20, driven by everything from catalyst promoters, glass amplifiers. Astatine, which has no stable isotopes, was first synthetically produced (1940) at the University of California by American physicists Dale R. The effective dose received in a typical chest X-ray examination is about 0.05 mSv. astatine (At), radioactive chemical element and the heaviest member of the halogen elements, or Group 17 (VIIa) of the periodic table. A radiation worker may legally receive up to 20 mSv per year. The annual dose received by a member of the public in Hong Kong from natural background radiation is about 2 mSv. This is a very large unit and submultiples, in particular, the millisievert (mSv - one thousandth of a sievert) are commonly used. The effective dose is frequently abbreviated to dose. Being a member of the alkali metals family of periodic table elements, caesium has been classified as one of the incompatible elements. Even though this element has a large number of isotopes, it has only one naturally occurring stable isotope, 133 Cs. It’s the 45th most abundant element in Earth’s crust. Caesium has at least 39 known isotopes, which is more than any other element except francium.The atomic masses of these isotopes range from 112 to 151. The effective dose is a single number broadly representing the risk to health taking into consideration the susceptibility to harm of different tissues and weighted for the harmfulness of different types of radiation. List of Elements A-Z Caesium (Cs) (Cesium) Caesium is a chemical element with an atomic number of 55 in the periodic table of elements. To put all ionising radiation on an equal basis with regard to the potential for causing harm, a quantity known as effective dose is introduced. Dose and Sievert - The effect of radiation on any material is determined by the "dose" of radiation that material receives. Calcium is used to produce the minerals contained in bones, shells and teeth through a process called biomineralisation. Which is what the old milk TV adverts were trying to tell us after all. Isotopes of an element can be classified as radioisotopes (those undergoing decay with the emission of characteristic radiation) or stable isotopes. Calcium is what is known as an essential element, meaning that it is an element which is absolutely necessary for life processes. 
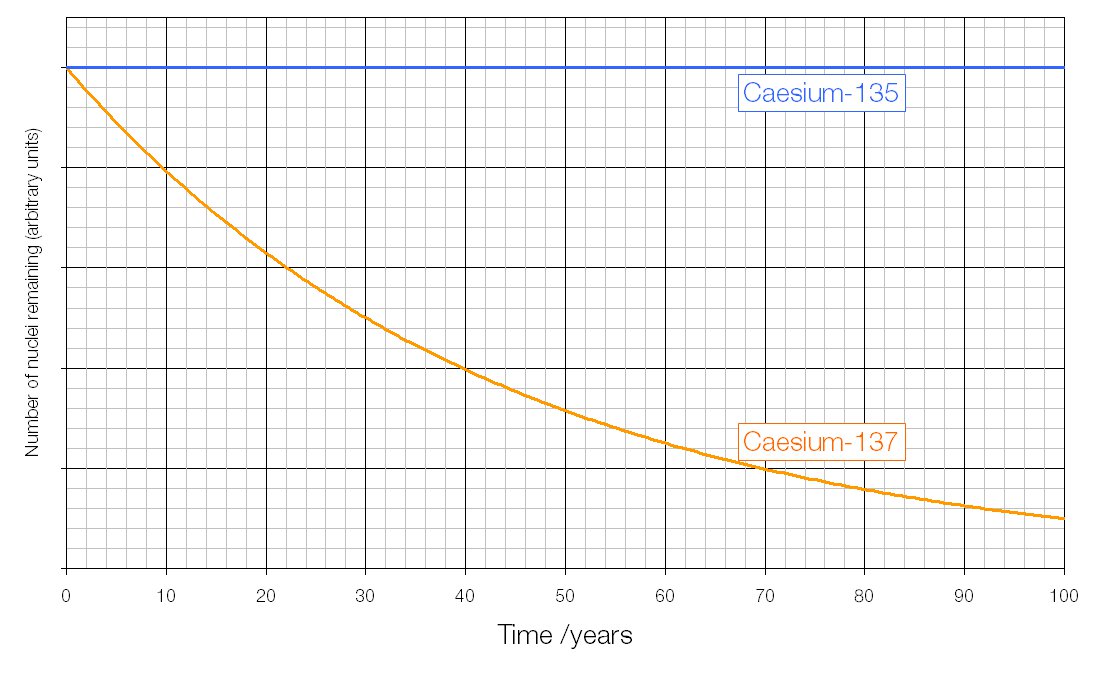
Different isotopes have the same chemical properties, but somewhat different physical properties. Atoms of an element having the same number of protons but different number of neutrons in the nuclei. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
